
Hydrogen has one valence electron, two bonded electrons, and zero unbonded electrons. The four hydrogens in this molecule are all identical, thus we can calculate all of their formal charges at once. Let’s see if this is true.īoron has three valence electrons, eight bonded electrons, and zero unbonded electrons. 
This means that the formal charges of all the individual atoms in it should add up to -1. 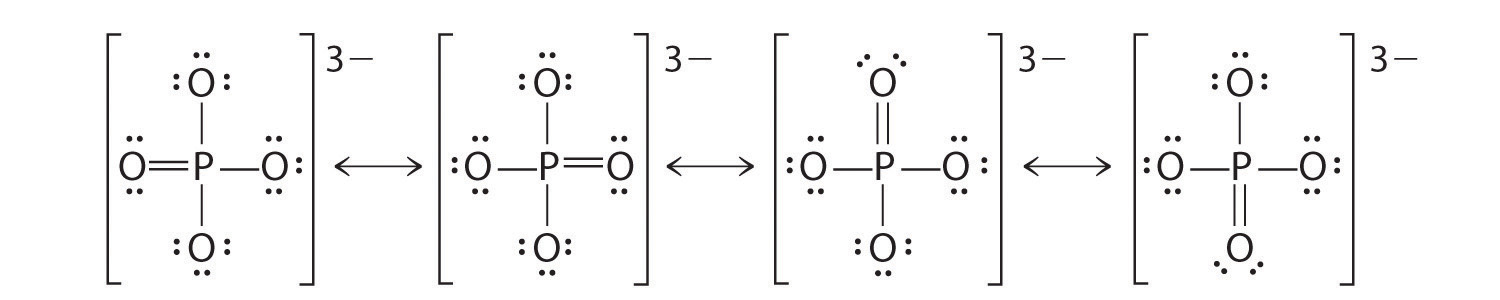
That is, they should sum up to zero if its an neutral molecule, and should sum up to the ion’s charge if it is not.Įxample: the ion BH 4 – has an overall charge of -1. It is also worth noting that the sum of all the formal charges of the atoms in a molecule must equal the overall charge on the molecule/ion. Oxygen is more electronegative than nitrogen, meaning the preferred structure is the one with a negative charge on the oxygen-the top structure! This leaves rule #4, meaning the preferred structure is the one with the negative charge on the more electronegative atom. Both the top and middle structures have adjacent atoms with opposite charges, so both satisfy rule #3. This eliminates the bottom structure, as it has a greater number of nonzero charges than the top two (it also has greater charges, as it contains a -2 charge, whereas the other two only contain +/-1). First, are there any structures possible where all the formal charges are zero? There are not, so we move on to rule #2. Given these calculated formal charges, let’s consult the guidelines discussed above. Next, the formal charge of the nitrogen in the middle: (5) – (½)(8) – (0) = +1.įinally, the formal charge of the oxygen: (6) – (½)(4) – (4) = 0.Īgain, first we calculate the formal charge of the nitrogen on the left: (5) – (½)(2) – (6) = -2.įinally, the formal charge of the oxygen: (6) – (½)(6) – (2) = +1. Similarly, we calculate the formal charge of the nitrogen on the left: (5) – (½)(4) – (4) = -1. Oxygen has 6 valence electrons, and this atom has 2 bonded electrons and 6 unbonded, thus the FC is (6) – (½)(2) – (6) = -1. This one has 8 bonded electrons and no unbonded, thus the FC is (5) – (½)(8) – (0) = +1.įinally, we calculate the formal charge of the oxygen. Next, we calculate it for the nitrogen in the middle. Nitrogen has 5 valence electrons, this atom has 6 bonded electrons (a triple bond), and 2 unbonded electrons, thus the formal charge is (5) – (½)(6) – (2) = 0. Let’s figure out which structure is correct.įirst, we calculate the formal charge of the nitrogen on the left.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
